(Reuters) -The U.S. Food and Drug Administration is probing the death of a patient who developed harmful antibodies after taking Takeda Pharmaceuticals' blood disorder therapy, the health regulator said on Friday.
The pediatric patient died about 10 months after starting Takeda's drug Adzynma as a preventive therapy, the agency said.
The child had congenital thrombotic thrombocytopenic purpura (cTTP), an inherited condition that causes blood clots in small vessels and can lead to organ damage.
The FDA said the child developed antibodies that blocked the activity of ADAMTS13, an enzyme critical for blood clotting.
Takeda did not immediately respond to Reuters request for comment.
Adzynma, approved in 2023 as the first therapy for cTTP, replaces the ADAMTS13 protein to help prevent dangerous blood clots.
The agency added it has received multiple postmarketing reports of patients developing neutralizing antibodies to ADAMTS13 after treatment with Adzynma.
(Reporting by Kamal Choudhury in Bengaluru; Editing by Vijay Kishore)
LATEST POSTS
- 1
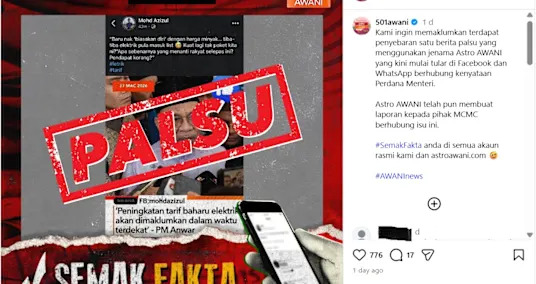
 Malaysian broadcaster rejects altered graphic about electricity rate hike
Malaysian broadcaster rejects altered graphic about electricity rate hike - 2
 Americans generally like wolves − except when we’re reminded of our politics
Americans generally like wolves − except when we’re reminded of our politics - 3
 Well known SUVs With Low Energy Utilization In 2024 vote
Well known SUVs With Low Energy Utilization In 2024 vote - 4
 'Stranger Things' made him a heartthrob. He left Hollywood anyway.
'Stranger Things' made him a heartthrob. He left Hollywood anyway. - 5
 The German series proving subtitles can be sexy — and wildly addictive
The German series proving subtitles can be sexy — and wildly addictive
 Pick the Ideal Family Feline Variety for Your Home
Pick the Ideal Family Feline Variety for Your Home 6 Well known Nissan Vehicles in the U.S.
6 Well known Nissan Vehicles in the U.S. Merz: 80% of Syrians in Germany should return in three years
Merz: 80% of Syrians in Germany should return in three years Looking for a great Thanksgiving side dish recipe? These are the crowd-pleasers the Yahoo team swears by.
Looking for a great Thanksgiving side dish recipe? These are the crowd-pleasers the Yahoo team swears by. A Lone Wolf Outsmarted Hunters in the Black Forest and Then Vanished
A Lone Wolf Outsmarted Hunters in the Black Forest and Then Vanished The 15 Most Compelling Books in History
The 15 Most Compelling Books in History Huge Iranian missile fragments, intercepted by air defenses, lay scattered across Israel, West Bank
Huge Iranian missile fragments, intercepted by air defenses, lay scattered across Israel, West Bank The Best 15 Applications for Efficiency and Association
The Best 15 Applications for Efficiency and Association Under pressure at home, Belgium's leader treads a tight rope with EU partners over funds for Ukraine
Under pressure at home, Belgium's leader treads a tight rope with EU partners over funds for Ukraine